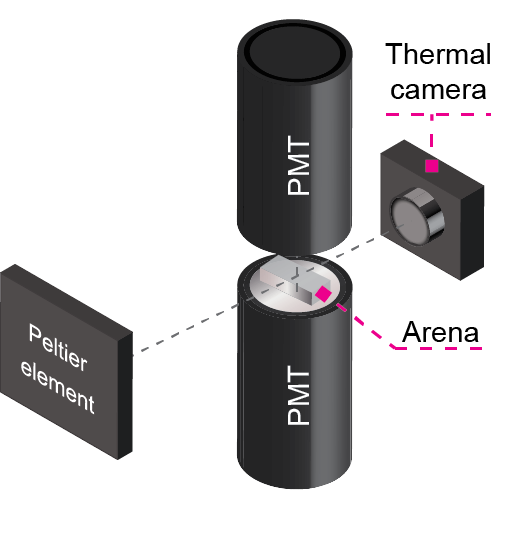
Many species from flies to mammals use signaling molecules called pheromones to attract mates and communicate socially. These pheromones may waft off the body or be excreted in urine, but now researchers at Japan’s RIKEN Center for Brain Science have found that pheromone-laced fecal landmarks, rather than flies’ bodies themselves, are strong attractors for fellow flies. In a small arena equipped for both visible light and thermal imaging, Drosophila flies could move around untethered. The researchers were interested in responses of both male and female flies to the male pheromone cVA, which activates only a specific class of odor-sensing neurons. In genetically-engineered flies, these neurons glow when activated thanks to the introduction of bioluminescent molecules similar to those in another familiar insect—fireflies.
“This simultaneous neural and behavioral recording is the first for investigating social communication in such small organisms,” observes research group leader Hokto Kazama. “The fly environment is a rich landscape of pheromones changing over space and time and this kind of naturalistic observation can change our understanding of what drives flies’ behavior.”
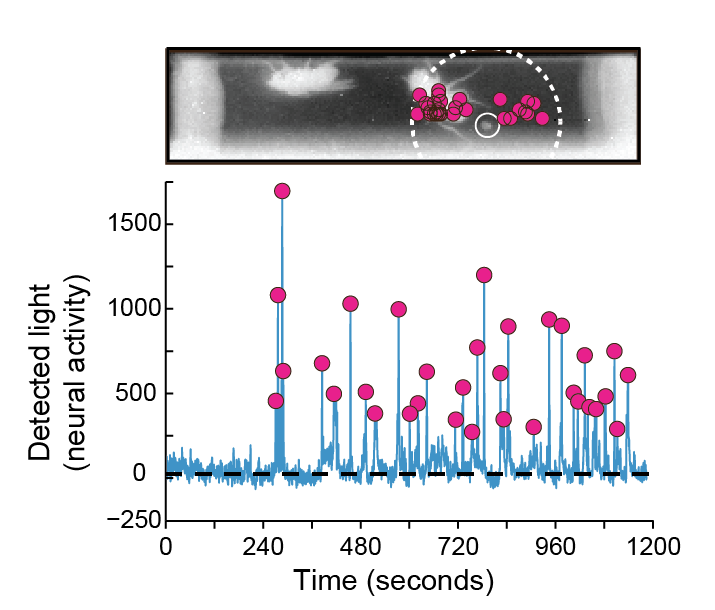
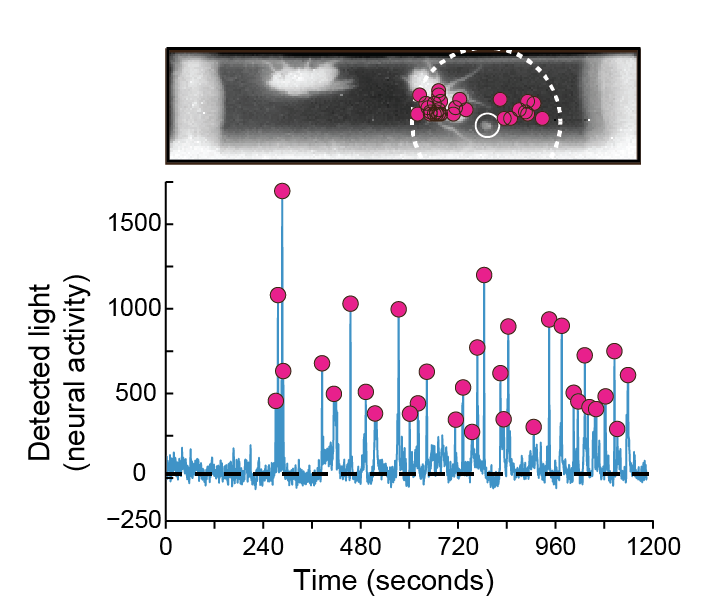
Observing male flies in the arena, the researchers found that odor-sensing neurons were triggered most strongly by a particular place—confirmed to be the location of a dropping—rather than interactions between flies, whose bodies were thought to be the main if not the sole source of cVA. “We have seen cases where the odor-sensing neurons respond only after a marking,” says Kazama, “and the closer the flies are to these fecal landmarks, the more bioluminescence we see.” On their own in the arena, male flies will circle the perimeter and deposit droppings, with a tendency to re-visit and spend more time around the marked regions. Flies with blocked odor-sensing neurons didn’t show this attraction.
Many species from flies to mammals use signaling molecules called pheromones to attract mates and communicate socially. These pheromones may waft off the body or be excreted in urine, but now researchers at Japan’s RIKEN Center for Brain Science have found that pheromone-laced fecal landmarks, rather than flies’ bodies themselves, are strong attractors for fellow flies. In a small arena equipped for both visible light and thermal imaging, Drosophila flies could move around untethered. The researchers were interested in responses of both male and female flies to the male pheromone cVA, which activates only a specific class of odor-sensing neurons. In genetically-engineered flies, these neurons glow when activated thanks to the introduction of bioluminescent molecules similar to those in another familiar insect—fireflies.
“This simultaneous neural and behavioral recording is the first for investigating social communication in such small organisms,” observes research group leader Hokto Kazama. “The fly environment is a rich landscape of pheromones changing over space and time and this kind of naturalistic observation can change our understanding of what drives flies’ behavior.”
Observing male flies in the arena, the researchers found that odor-sensing neurons were triggered most strongly by a particular place—confirmed to be the location of a dropping—rather than interactions between flies, whose bodies were thought to be the main if not the sole source of cVA. “We have seen cases where the odor-sensing neurons respond only after a marking,” says Kazama, “and the closer the flies are to these fecal landmarks, the more bioluminescence we see.” On their own in the arena, male flies will circle the perimeter and deposit droppings, with a tendency to re-visit and spend more time around the marked regions. Flies with blocked odor-sensing neurons didn’t show this attraction.
In another experiment with a male and female fly, the female’s droppings were not attractive. The male’s markings, on the other hand, were attractive to both sexes, even during courtship when a female is chased by a male. Fecal landmarks thus function as a social hub, indicating the identity of the marking fly and increasing mating chances.
The system for monitoring bioluminescence has applications beyond fruit flies. Any neural recording, especially of small animals, that would traditionally require wires or tethering can now harness the power of these glowing brains, giving researchers a real-time look at activity during social communication.
“Emerging genome editing technologies combined with bioluminescent probes are giving us a powerful window into observing natural behavior and how it arises in the brain,” says Kazama. ✅
Amanda Alvarez ? is a science writer at the RIKEN Center for Brain Science. Read more about this study on her Neurographics blog
In another experiment with a male and female fly, the female’s droppings were not attractive. The male’s markings, on the other hand, were attractive to both sexes, even during courtship when a female is chased by a male. Fecal landmarks thus function as a social hub, indicating the identity of the marking fly and increasing mating chances.
The system for monitoring bioluminescence has applications beyond fruit flies. Any neural recording, especially of small animals, that would traditionally require wires or tethering can now harness the power of these glowing brains, giving researchers a real-time look at activity during social communication.
“Emerging genome editing technologies combined with bioluminescent probes are giving us a powerful window into observing natural behavior and how it arises in the brain,” says Kazama. ✅
Amanda Alvarez ? is a science writer at the RIKEN Center for Brain Science. Read more about this study on her Neurographics blog
Mercier D, Tsuchimoto Y, Ohta K, Kazama H (2018) Olfactory landmark-based communication in interacting Drosophila. Current Biology. doi: 10.1016/j.cub.2018.06.005