In books and the movies, a group of people on a special mission always sends out a scout to do reconnaissance before they proceed. Sometimes, the scouts leave signs or markers that allow the group to know where there should go. Researchers led by Azusa Inoue at the RIKEN Center for Integrative Medical Sciences (IMS) in Japan have discovered a mark left behind in unfertilized egg cells that determine which DNA modifications will be inherited if the egg is fertilized. Specifically, they found that without initial modifications to histone H2A at lysine 119—technically called H2AK119ub1—later inheritable modifications would not occur. When allowed to develop, one consequence of this deficit was an enlarged placenta after embryo implantation. This study was published in Nature Genetics on April 5.
For many years we were taught in school that acquired traits were not inherited. In some sense this was correct; stretching your neck a lot to get food will not result in children with longer necks. However, your DNA function can be modified throughout your life. For example, DNA structure in chromosomes is supported by proteins called histones. When histones are modified, they can change how genes are expressed in the body. This is epigenetics, and a previous study by Inoue and colleagues showed that acquired tri-methylation of histone H3 at lysine 27 (thankfully abbreviate to H3K27me3) in mammalian egg cells can be inherited. In the new study, the team used technology called low-input CUT&RUN to begin answering the question of how this happens.
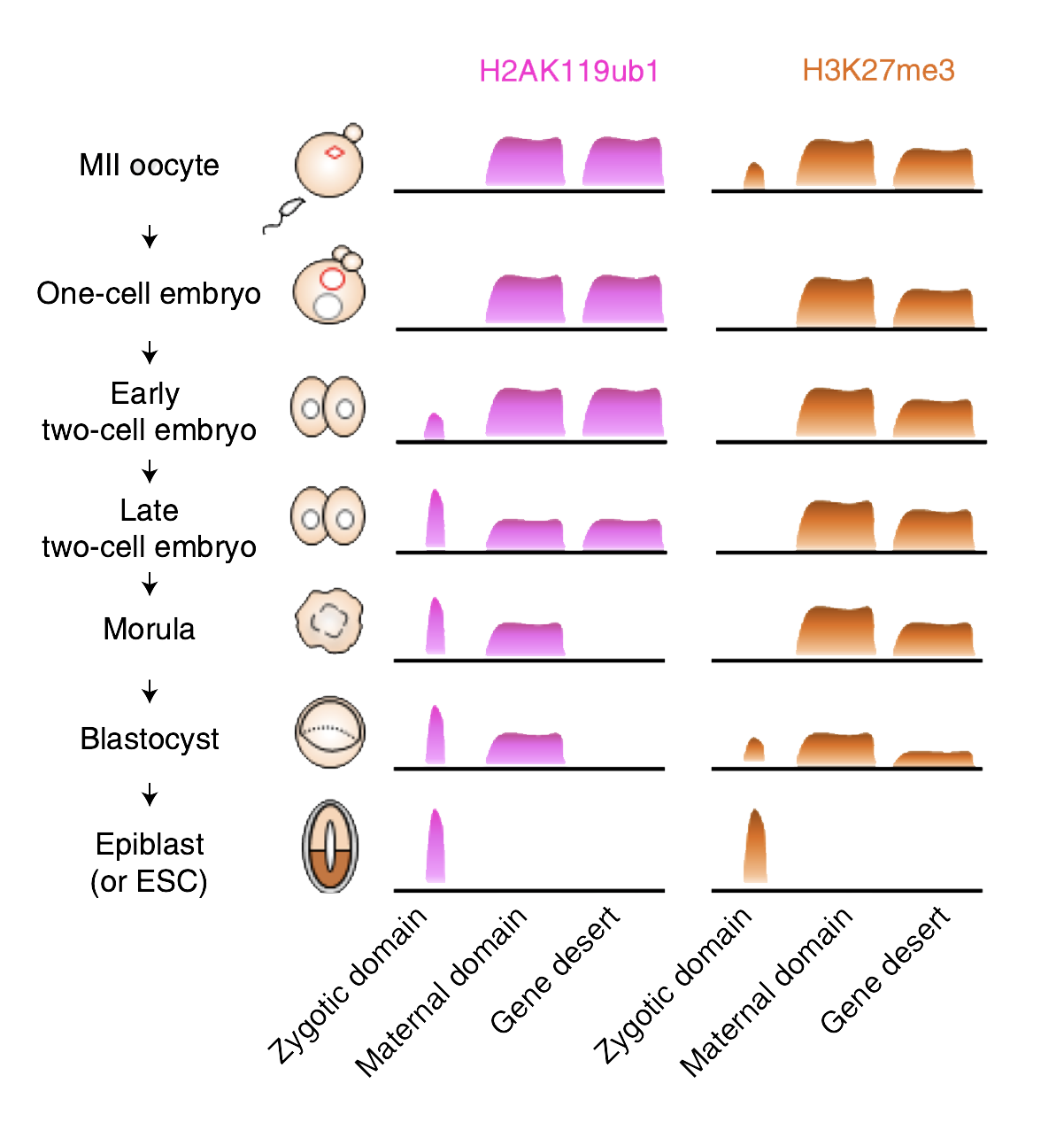
Schematic of the temporal dynamics of H2AK119ub1 and H3K27me3 during the maternal-to-zygotic transition. (click to expand)
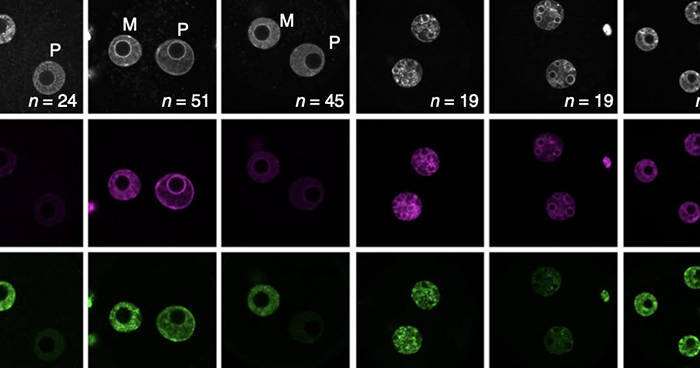
First, the researchers examined the timing of the two different histone modifications. They found that every gene exhibiting H3K27me3 also showed H2AK119ub1 in mouse egg cells. Suspecting its importance, the researchers knocked out two proteins that make up H2AK119ub1 in egg cells. Low-input CUT&RUN showed that the knock-out egg cells had much less H3K27me3 than controls at a subset of genes that normally bring H3K27me3 into the next generation.
Thus, H2AK119ub1 acts like a kind of marker left by a scout, identifying where subsequent H3K27me3 should follow. “We discovered that H2AK119ub1 is necessary for maternal inheritance of H3K27me3, making the H2AK119ub1-H3K27me3 pathway a major player in transgenerational epigenetic inheritance in mammals,” says Inoue.
The researchers then found something they didn’t expect. Testing showed that the loss of H3K27me3 was itself inherited by fertilized embryos, and could not be reversed. Furthermore, this deficiency led increased lethality—miscarriages—and enlarged placentas. “It was surprising to find that defects in an egg’s histone modification are irreversibly inherited by embryos and cause long term consequences in development,” says Inoue.
The results thus showed that despite normal DNA in the mouse egg cell, if the proper instructions—first H2AK119ub1 and then H3K27me3 modifications—were missing, miscarriages and enlarged placentas could occur. These findings have clinical implications, especially for reproductive medicine and placental defects. “The next step,” says Inoue, “is to see whether any diseases or surrounding environments can affect the heritable histone modification.” ✅
For many years we were taught in school that acquired traits were not inherited. In some sense this was correct; stretching your neck a lot to get food will not result in children with longer necks. However, your DNA function can be modified throughout your life. For example, DNA structure in chromosomes is supported by proteins called histones. When histones are modified, they can change how genes are expressed in the body. This is epigenetics, and a previous study by Inoue and colleagues showed that acquired tri-methylation of histone H3 at lysine 27 (thankfully abbreviate to H3K27me3) in mammalian egg cells can be inherited. In the new study, the team used technology called low-input CUT&RUN to begin answering the question of how this happens.
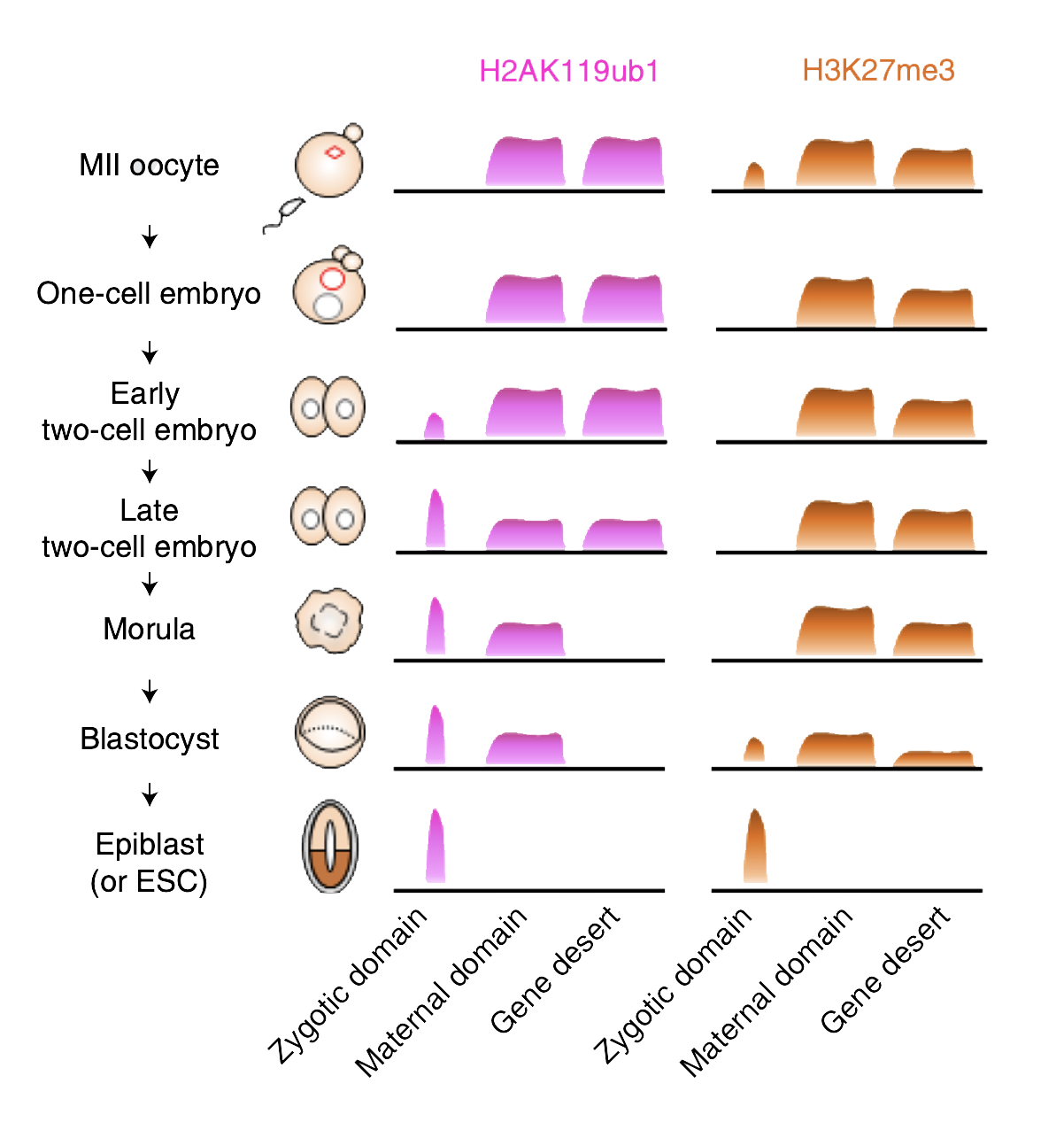
Schematic of the temporal dynamics of H2AK119ub1 and H3K27me3 during the maternal-to-zygotic transition. (click to expand)
First, the researchers examined the timing of the two different histone modifications. They found that every gene exhibiting H3K27me3 also showed H2AK119ub1 in mouse egg cells. Suspecting its importance, the researchers knocked out two proteins that make up H2AK119ub1 in egg cells. Low-input CUT&RUN showed that the knock-out egg cells had much less H3K27me3 than controls at a subset of genes that normally bring H3K27me3 into the next generation.
Thus, H2AK119ub1 acts like a kind of marker left by a scout, identifying where subsequent H3K27me3 should follow. “We discovered that H2AK119ub1 is necessary for maternal inheritance of H3K27me3, making the H2AK119ub1-H3K27me3 pathway a major player in transgenerational epigenetic inheritance in mammals,” says Inoue.
The researchers then found something they didn’t expect. Testing showed that the loss of H3K27me3 was itself inherited by fertilized embryos, and could not be reversed. Furthermore, this deficiency led increased lethality—miscarriages—and enlarged placentas. “It was surprising to find that defects in an egg’s histone modification are irreversibly inherited by embryos and cause long term consequences in development,” says Inoue.
The results thus showed that despite normal DNA in the mouse egg cell, if the proper instructions—first H2AK119ub1 and then H3K27me3 modifications—were missing, miscarriages and enlarged placentas could occur. These findings have clinical implications, especially for reproductive medicine and placental defects. “The next step,” says Inoue, “is to see whether any diseases or surrounding environments can affect the heritable histone modification.” ✅
Further reading
Mei H, Kozuka C, Hayashi R, Kumon M, Koseki H, Inoue A (2021) H2AK119ub1 guides maternal inheritance and zygotic deposition of H3K27me3 in mouse embryos. Nat. Genet. doi: 10.1038/s41588-021-00820-3
Further reading
Mei H, Kozuka C, Hayashi R, Kumon M, Koseki H, Inoue A (2021) H2AK119ub1 guides maternal inheritance and zygotic deposition of H3K27me3 in mouse embryos. Nat. Genet. doi: 10.1038/s41588-021-00820-3