Aya Takeoka at the RIKEN Center for Brain Science (CBS) in Japan and colleagues at Neuro-Electronics Research Flanders (NERF, empowered by IMEC, KU Leuven, and VIB) have discovered have discovered the neural circuitry in the spinal cord that allows brain-independent motor learning. Recently published in Science, the study found two critical groups of spinal cord neurons, one necessary for new adaptive learning, and another for recalling adaptations once they have been learned. The findings could help scientists develop ways to assist motor recovery after spinal cord injury.
Scientists have known for some time that motor output from the spinal cord can be adjusted through practice even without a brain. This has been shown most dramatically in headless insects, whose legs can still be trained to avoid external cues. Until now, no one has figured out exactly how this is possible, and without this understanding, the phenomenon is not much more than a quirky fact. As Takeoka explains, “Gaining insights into the underlying mechanism is essential if we want to understand the foundations of movement automaticity in healthy people and use this knowledge to improve recovery after spinal cord injury.”
Before jumping into the neural circuitry, the researchers first developed an experimental setup that allowed them to study mouse spinal cord adaptation, both learning and recall, without input from the brain. Each test had an experimental mouse and a control mouse whose hindlegs dangled freely. If the experimental mouse’s hindleg drooped down too much it was electrically stimulated, emulating something a mouse would want to avoid. The control mouse received the same stimulation at the same time, but not linked to its own hindleg position.
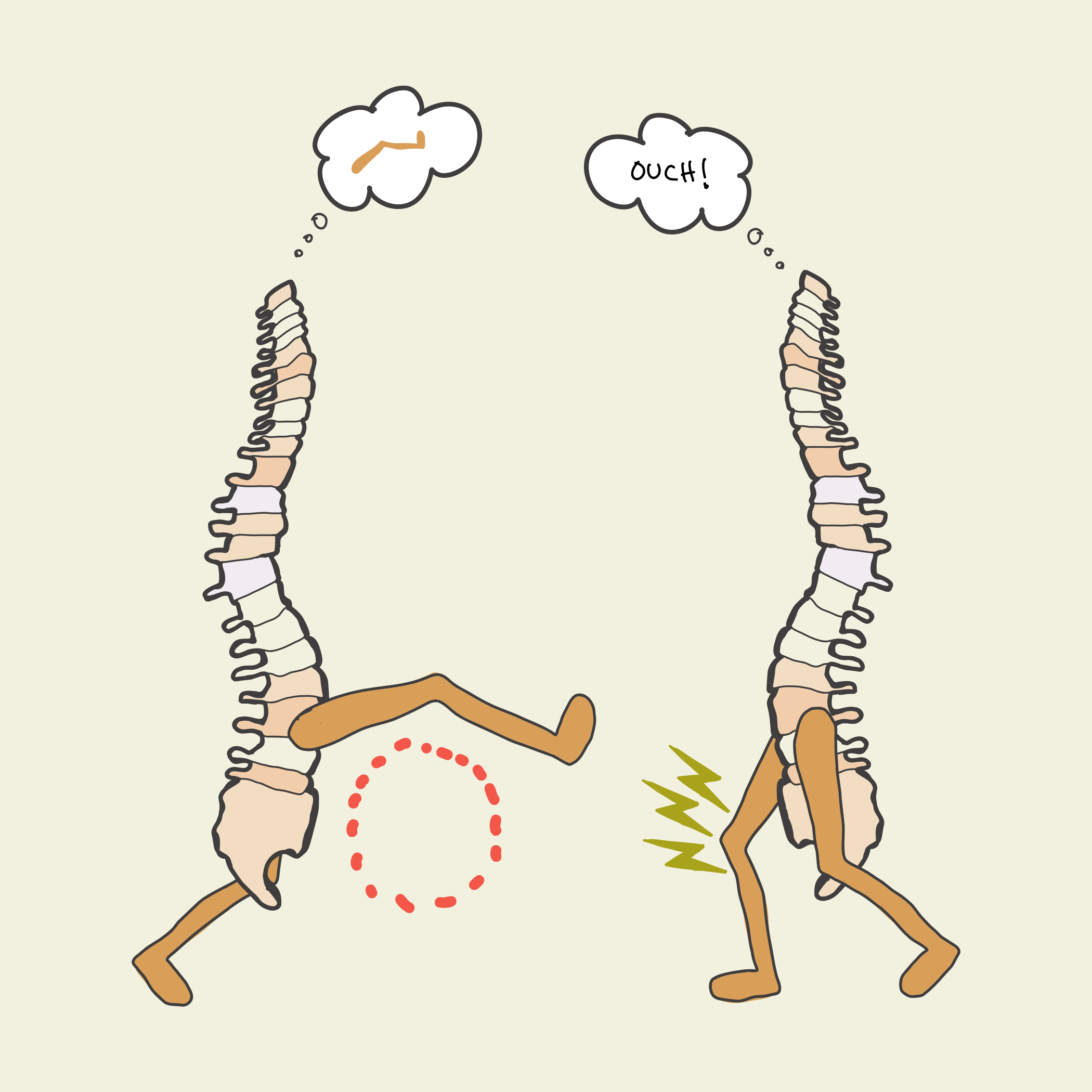
In this study, spinal cords that associated limb position with an unpleasant experience (left) learned to reposition the limb after only 10 minutes, and retained a memory the next day. Spinal cords that received random unpleasantness (right) did not learn.
After just 10 minutes, they observed motor learning only in the experimental mice; their legs remained high up, avoiding any electrical stimulation. This result showed that the spinal cord can associate an unpleasant feeling with leg position and adapt its motor output so that the leg avoids the unpleasant feeling, all without any need for a brain. Twenty-four hours later, they repeated the 10-minute test but reversed the experimental and control mice. The original experimental mice still kept their legs up, indicating that the spinal cord retained a memory of the past experience, which interfered with new learning.
Having thus established both immediate learning, as well as memory, in the spinal cord, the team then set out to examine the neural circuitry that makes both possible. They used six types of transgenic mice, each with a different set of spinal neurons disabled, and tested them for motor learning and learning reversal. They found that mice hindlimbs did not adapt to avoid the electrical shocks after neurons toward the top of the spinal cord were disabled, particularly those that express the gene Ptf1a.
When they examined the mice during learning reversal, they found that silencing the Ptf1a-expressing neurons had no effect. Instead, a group of neurons in the bottom, ventral, part of the spinal cord that express the En1 gene was critical. When these neurons were silenced the day after learning avoidance, the spinal cords acted as if they had never learned anything. The researchers also assessed memory recall on the second day by repeating the initial learning conditions. They found that in wildtype mice, hindlimbs stabilized to reach the avoidance position faster than they did on the first day, indicating recall. Exciting the En1 neurons during recall increased this speed by 80%, indicating enhanced motor recall.
“Not only do these results challenge the prevailing notion that motor learning and memory are solely confined to brain circuits,” says Takeoka, “but we showed that we could manipulate spinal cord motor recall, which has implications for therapies designed to improve recovery after spinal cord damage.”
Scientists have known for some time that motor output from the spinal cord can be adjusted through practice even without a brain. This has been shown most dramatically in headless insects, whose legs can still be trained to avoid external cues. Until now, no one has figured out exactly how this is possible, and without this understanding, the phenomenon is not much more than a quirky fact. As Takeoka explains, “Gaining insights into the underlying mechanism is essential if we want to understand the foundations of movement automaticity in healthy people and use this knowledge to improve recovery after spinal cord injury.”
Before jumping into the neural circuitry, the researchers first developed an experimental setup that allowed them to study mouse spinal cord adaptation, both learning and recall, without input from the brain. Each test had an experimental mouse and a control mouse whose hindlegs dangled freely. If the experimental mouse’s hindleg drooped down too much it was electrically stimulated, emulating something a mouse would want to avoid. The control mouse received the same stimulation at the same time, but not linked to its own hindleg position.
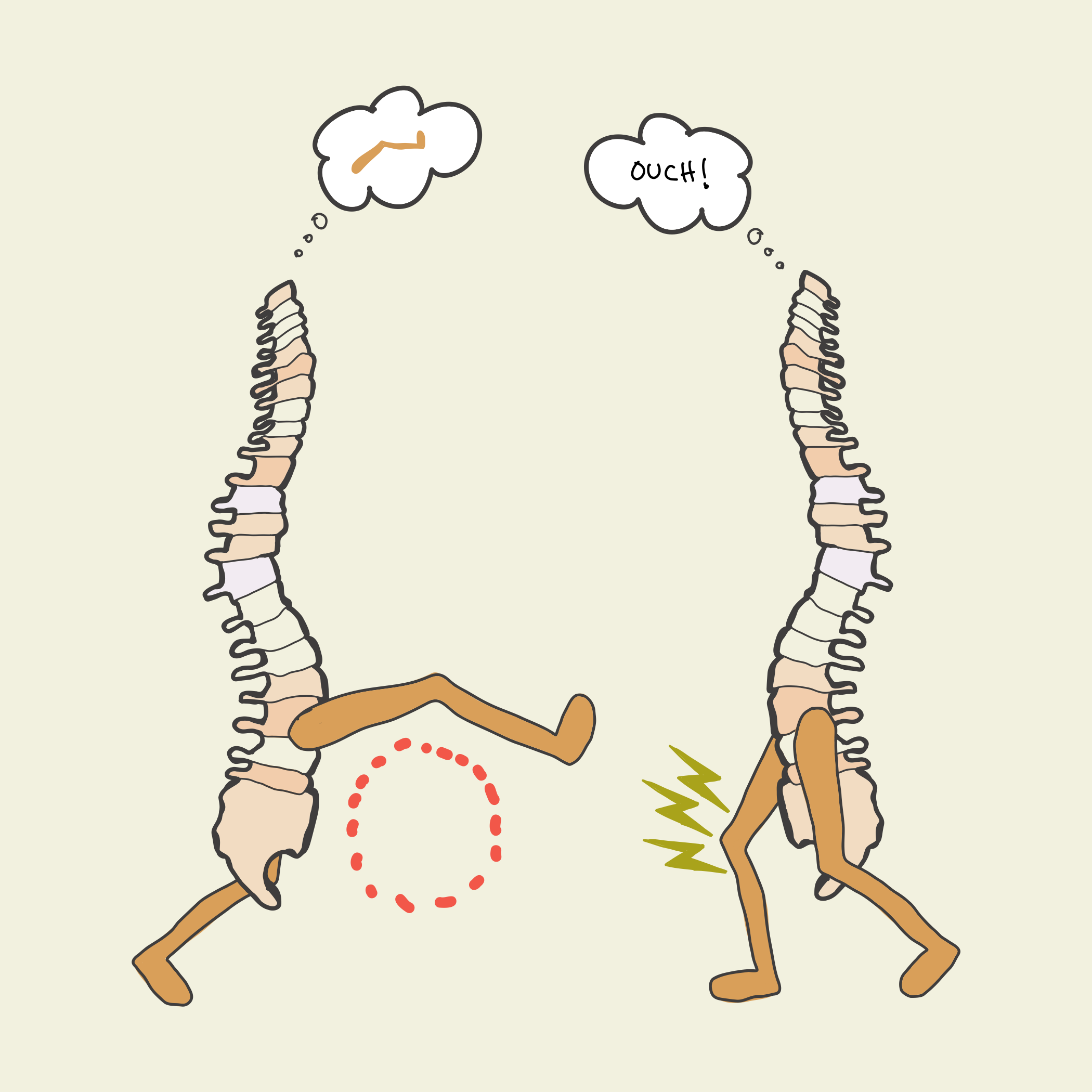
In this study, spinal cords that associated limb position with an unpleasant experience (left) learned to reposition the limb after only 10 minutes, and retained a memory the next day. Spinal cords that received random unpleasantness (right) did not learn.
After just 10 minutes, they observed motor learning only in the experimental mice; their legs remained high up, avoiding any electrical stimulation. This result showed that the spinal cord can associate an unpleasant feeling with leg position and adapt its motor output so that the leg avoids the unpleasant feeling, all without any need for a brain. Twenty-four hours later, they repeated the 10-minute test but reversed the experimental and control mice. The original experimental mice still kept their legs up, indicating that the spinal cord retained a memory of the past experience, which interfered with new learning.
Having thus established both immediate learning, as well as memory, in the spinal cord, the team then set out to examine the neural circuitry that makes both possible. They used six types of transgenic mice, each with a different set of spinal neurons disabled, and tested them for motor learning and learning reversal. They found that mice hindlimbs did not adapt to avoid the electrical shocks after neurons toward the top of the spinal cord were disabled, particularly those that express the gene Ptf1a.
When they examined the mice during learning reversal, they found that silencing the Ptf1a-expressing neurons had no effect. Instead, a group of neurons in the bottom, ventral, part of the spinal cord that express the En1 gene was critical. When these neurons were silenced the day after learning avoidance, the spinal cords acted as if they had never learned anything. The researchers also assessed memory recall on the second day by repeating the initial learning conditions. They found that in wildtype mice, hindlimbs stabilized to reach the avoidance position faster than they did on the first day, indicating recall. Exciting the En1 neurons during recall increased this speed by 80%, indicating enhanced motor recall.
“Not only do these results challenge the prevailing notion that motor learning and memory are solely confined to brain circuits,” says Takeoka, “but we showed that we could manipulate spinal cord motor recall, which has implications for therapies designed to improve recovery after spinal cord damage.”
Further reading
Lavaud S, Bichara C, D’Andola M, Yeh S, and Takeoka A (2024) Two inhibitory neuronal classes govern acquisition and recall of spinal sensorimotor adaptation. Science. doi: 10.1126/science.adf6801
Further reading
Lavaud S, Bichara C, D’Andola M, Yeh S, and Takeoka A (2024) Two inhibitory neuronal classes govern acquisition and recall of spinal sensorimotor adaptation. Science. doi: 10.1126/science.adf6801