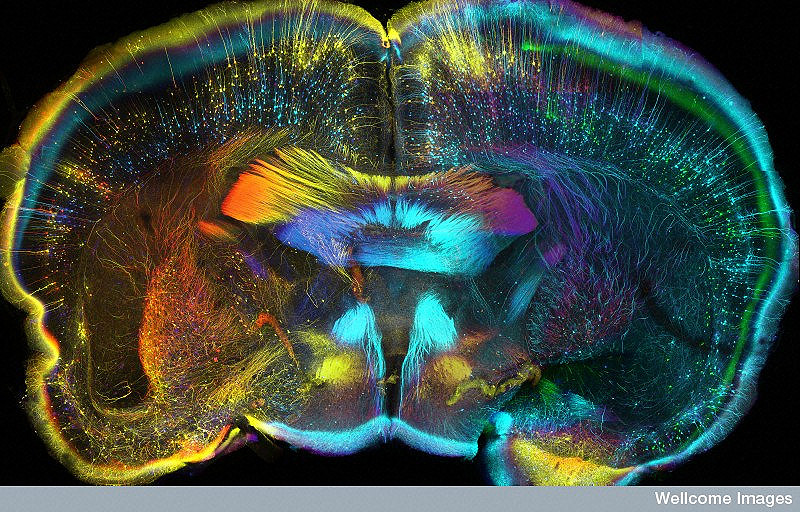
A coronal view of a mouse brain. Image: Wellcome Images, CC BY-NC-ND 2.0
This post originally appeared on Medium
Like many people, mice are suckers for novelty. They will explore new environments and objects more than familiar ones, and this preference for novelty is the basis for memory testing in mice. Given the choice, if mice spend more time examining new surroundings, the reasoning goes, it’s because they remember places and things they’ve already been exposed to. But if the right neurons don’t get turned on during sleep, mice don’t remember what they’ve experienced and treat the familiar as if it were new. The parts of the brain responsible for creating the memory itself — the sensory and motor cortex areas that mediate the feeling of exploration — must be re-activated during a specific part of the sleep cycle for the mouse to remember.
It’s no surprise that, for both humans and mice, sleep is pretty important for learning and remembering. What wasn’t known was that higher brain regions, as opposed to canonical memory areas like the hippocampus, participate in consolidating memories. In an intricate set of experiments, researchers in Japan either activated or silenced motor and sensory cortical areas in mice and then looked at how well they remembered.

A mouse exploring the two floor textures during the experiment. Image: Masanori Murayama/RIKEN Brain Science Institute
The study started with mice experiencing a new environment with either smooth or grooved floors. Not surprisingly, when mice were given a choice one day later, they preferred the previously unexplored texture. The brain areas mediating the mouse’s feeling of the floor and the associated movement are reciprocally connected: touch input from the paw goes from primary somatosensory cortex (S1) to secondary motor cortex (M2), which in turn projects ‘top-down’ back to S1. This sensorimotor circuit was the target of a memory manipulation, achieved using optogenetics ([itg-tooltip tooltip-content=”<p><img src=&aquot;http://www.scientificamerican.com/media/promo/upload/MiQ511Cart.gif&aquot; /></p>”]here’s a simple[/itg-tooltip] intro to this method of controlling neurons with light).
After 10 minutes of exploring, mice took a rest, and this is where the optogenetics comes in. The researchers independently turned off M2 or S1 with light during different brain states corresponding to wakefulness or non-REM (rapid eye movement) sleep. If the top-down signal from M2 was silenced, in a later memory test mice had ‘forgotten’ the learned environment, but only if M2 was turned off early in the sleep cycle during the non-REM phase. Switching off M2 while the mouse was awake and resting, or switching off S1 during non-REM sleep, did not have any impact on mouse memory.
The authors were able to achieve even more potent control of memory, again with optogenetics. The memories of mice could effectively be wiped out in a day, or prolonged for up to four days, depending on the synchrony of light stimulation to S1 and M2. Simultaneous stimulation caused neurons in the two cortical areas to activate more during non-REM sleep. The optogenetic control of neurons created a synchrony of slow brain wave activity between S1 and M2 that is typical of deep, non-REM sleep. Even sleep-deprived mice retained their memory of the explored environment when they were given simultaneous light stimulation to both S1 and M2.
Turning on the same neurons that were active during the memory formation, when the mouse was exploring, thus seems to be important for retaining the memory in mice, at least for this texture learning task. The fact that this consolidation happens during sleep means brain stimulation techniques like transcranial magnetic stimulation or transcranial direct current stimulation applied at the right time could rescue memories in sleep disordered individuals, a direction the researchers are pursuing next.
This post originally appeared on Medium
Like many people, mice are suckers for novelty. They will explore new environments and objects more than familiar ones, and this preference for novelty is the basis for memory testing in mice. Given the choice, if mice spend more time examining new surroundings, the reasoning goes, it’s because they remember places and things they’ve already been exposed to. But if the right neurons don’t get turned on during sleep, mice don’t remember what they’ve experienced and treat the familiar as if it were new. The parts of the brain responsible for creating the memory itself — the sensory and motor cortex areas that mediate the feeling of exploration — must be re-activated during a specific part of the sleep cycle for the mouse to remember.
It’s no surprise that, for both humans and mice, sleep is pretty important for learning and remembering. What wasn’t known was that higher brain regions, as opposed to canonical memory areas like the hippocampus, participate in consolidating memories. In an intricate set of experiments, researchers in Japan either activated or silenced motor and sensory cortical areas in mice and then looked at how well they remembered.

A mouse exploring the two floor textures during the experiment. Image: Masanori Murayama/RIKEN Brain Science Institute
The study started with mice experiencing a new environment with either smooth or grooved floors. Not surprisingly, when mice were given a choice one day later, they preferred the previously unexplored texture. The brain areas mediating the mouse’s feeling of the floor and the associated movement are reciprocally connected: touch input from the paw goes from primary somatosensory cortex (S1) to secondary motor cortex (M2), which in turn projects ‘top-down’ back to S1. This sensorimotor circuit was the target of a memory manipulation, achieved using optogenetics ([itg-tooltip tooltip-content=”<p><img src=&aquot;http://www.scientificamerican.com/media/promo/upload/MiQ511Cart.gif&aquot; /></p>”]here’s a simple[/itg-tooltip] intro to this method of controlling neurons with light).
After 10 minutes of exploring, mice took a rest, and this is where the optogenetics comes in. The researchers independently turned off M2 or S1 with light during different brain states corresponding to wakefulness or non-REM (rapid eye movement) sleep. If the top-down signal from M2 was silenced, in a later memory test mice had ‘forgotten’ the learned environment, but only if M2 was turned off early in the sleep cycle during the non-REM phase. Switching off M2 while the mouse was awake and resting, or switching off S1 during non-REM sleep, did not have any impact on mouse memory.
The authors were able to achieve even more potent control of memory, again with optogenetics. The memories of mice could effectively be wiped out in a day, or prolonged for up to four days, depending on the synchrony of light stimulation to S1 and M2. Simultaneous stimulation caused neurons in the two cortical areas to activate more during non-REM sleep. The optogenetic control of neurons created a synchrony of slow brain wave activity between S1 and M2 that is typical of deep, non-REM sleep. Even sleep-deprived mice retained their memory of the explored environment when they were given simultaneous light stimulation to both S1 and M2.
Turning on the same neurons that were active during the memory formation, when the mouse was exploring, thus seems to be important for retaining the memory in mice, at least for this texture learning task. The fact that this consolidation happens during sleep means brain stimulation techniques like transcranial magnetic stimulation or transcranial direct current stimulation applied at the right time could rescue memories in sleep disordered individuals, a direction the researchers are pursuing next.
Reference:
Topics: ・biology・brain science・
neuroamanda
Science writer at the RIKEN Brain Science Institute in Japan
Blogging about brains on Medium