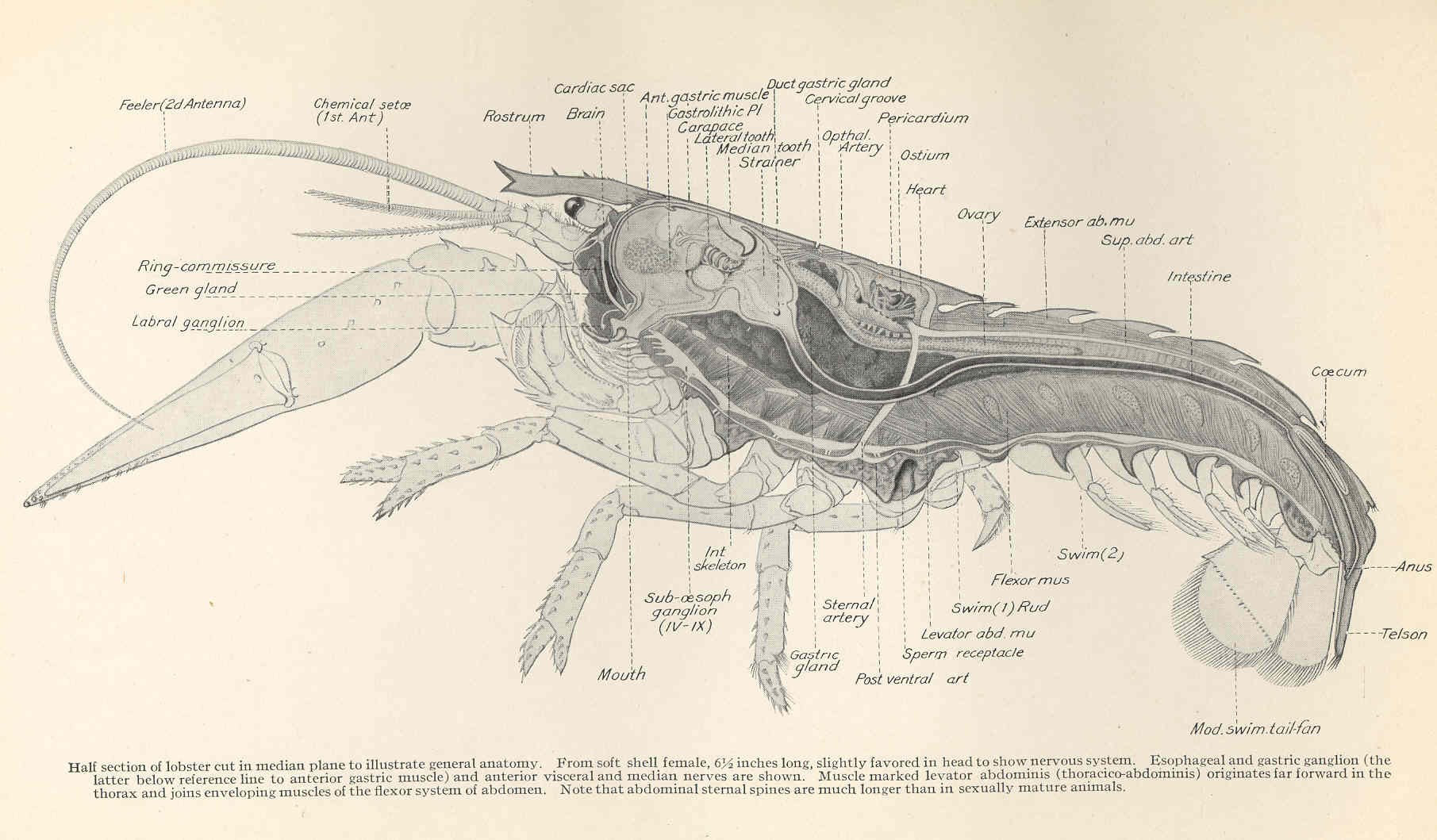
Anatomy of a lobster, a model organism studied by Eve Marder’s group. Image: Francis Hobart Herrick, Freshwater and Marine Image Bank via Wikimedia Commons, public domain
This post originally appeared on Medium
None of us would get on a plane that had its parts changed in mid-air, says Eve Marder, who has spent her career probing a very specific cluster of crustacean nerve cells. Yet we are all walking around undergoing a constant turnover of cellular parts, and so are the lobsters and crabs Marder studies. Within every nervous system, crucial components that mediate neural communication — receptors that detect chemical signals or pores that let ions in and out of neurons — are replaced hourly or daily, and the communication itself can be routed through various neurons. Marder studies how this complexity is maintained by artificially ‘crashing’ the nervous system.

The pyloric circuit, part of the group of nerve cells in the lobster that control digestion. The communication between these neurons is controlled by two neurotransmitters, acetylcholine (ACh in pink) and glutamate (Glu in blue). Image from Figure 4a of Marder & Bucher, 2007.
The clutch of lobster stomatogastric, or digestive, neurons is an ideal testbed for nervous system hacking: the small number of cells naturally maintain an exquisite temporal synchrony with each other but can be exposed to environmental extremes or modulated to produce configurations that drive the system to collapse. The orderly pattern of neural discharges then gives way to freakish rhythms or utter silence; these crashes reveal not just the variability that exists across cells and experimental animals but highlight the incredible robustness of the nervous system to external and internal shocks. Like an overheating laptop, the small network of cells can also shut down in high temperatures, though unlike a fried computer the cells can recover when the environment returns to normal.
How do nervous systems bounce back and maintain normal outputs in the face of constant internal change? This is the broader, career-spanning question for Marder, but her enthusiasm for hacking systems also extends from circuits to scientific publishing and beyond. During a recent visit to the RIKEN Brain Science Institute in Japan, the Brandeis professor and Kavli Prize winner talked to us about redundancy in the nervous system, her passion for open-access publishing, and why individual investigator grants are the way of the future.
“Connectomes are absolutely necessary but completely insufficient for understanding nervous system function”
How do people, or lobsters, maintain robust and functional behaviors while their nervous systems are constantly renewing and reconfiguring themselves?
Anything important the brain does, there will be more than one way of doing it. Circuit diagrams or connectomes are absolutely necessary but completely insufficient for understanding nervous system function. By studying degenerate mechanisms that produce the same behavior change we can probe the underlying parameter space, and I think this is a paradigm shift. We are not looking for one model that represents a mean of parameters. Rather, we try to build many models that cover the range of normal behavior parameters. The networks that we build may produce the same output with different parameter sets, but respond differently to perturbations. This kind of variability in animals is often hidden by compensating cellular mechanisms. How you can maintain a nervous system that is constantly breaking down and changing its parts, that for me is the deeper question.
“Knowledge should not be for sale”
As a Senior Editor at eLife, what does open access mean to you and are you seeing a shift, in both academics’ publishing choices and how peer review is conducted?
eLife is a rigorous journal that publishes roughly 15% of submitted papers, but we also have a philosophical stance on open access. For me personally, this came from my mother, who was interested in human brain development and in her later years was very deaf and consequently spent a lot of time online. She told me that academics forget that they have access, through their libraries, to all the world’s published research. But my mother, at home, didn’t have a university library. She said it’s criminal that she had to pay to access papers, because the abstract alone can’t tell you if you really want to read the paper or not. Fundamentally, knowledge should not be for sale. Most research, wherever it’s done, is funded through taxes, either directly by a government or indirectly through tax-privileged foundations and institutions. Working people, tax-paying people, should have rights to knowledge, and I feel strongly about that. Personally I will only publish in journals that have open-access options. Even commercial journals have options to make papers free six or twelve months after publication. Knowledge should not be privileged.
Researchers should also not be assessed based on impact factor — I know recent recruits who have gotten faculty jobs on the basis of papers in eLife or Journal of Neuroscience. Randy Schekman, eLife editor-in-chief, refuses to publicize eLife’s impact factor, though it is calculated by Web of Science, and it is quite respectable. eLife is a generalist journal and it has become a home for extraordinary papers that will become longstanding classics in their fields, such as the Drosophila lines or Homo naledi, because there is no limit on the number of figures or pages. These are papers with scope that some high-profile journals might be unwilling to publish. Some authors are sending their papers to eLife for other reasons, because they prefer the faster editorial process and know that we don’t ask for months of additional work to get a paper accepted.
High-profile journals have taught authors to oversell. This is the most common complaint I hear from reviewers: authors over-claiming novelty or generality, trying to make their result more important than it is.
“Brain tech development will make possible things that we could never have dreamed of”
How is the funding landscape changing for neuroscience?
I’ve been fortunate to have been funded by the NIH (US National Institutes of Health) throughout my career. The National Institute of Neurological Disorders and Stroke (NINDS) recently created a new grant mechanism,R35, which allows an investigator to consolidate several grants into a single multi-year project. Previously, in the US and working under NIH funding, you were tied to a specific project, and grants were awarded to projects not people. You had to keep strict track of which experiments belonged to which grant. This new award gives grants to people and is less restrictive. The model is more akin to that of the Howard Hughes Medical Institute or MacArthur Fellowships. When this change comes to pass, it will be a great relief for investigators to go in the direction the science takes us.
You were a member of the original BRAIN Initiative working group. What do you think the outlook is for neuroscience as the field becomes more global and collaborative?
The outlook is really positive. I’m not involved in the BRAIN Initiative anymore, but it was exciting to have been a part of its beginning, with 14 other very smart people who were all thinking unselfishly out of the box about what is best for the field. Everyone agreed that at that moment, new technologies that could be used by any investigator would be most useful, and that the best strategy was individual investigator awards and a bottom-up rather than top-down approach.
Last year at SfN, there were a number of talks from the first round of awards. Some really beautiful science was presented with interesting directions. This heyday of technology development is going to make possible things we could never have dreamed of. But at some point you have to transition from technology development to implementation. You may be recording from many neurons, but at the end of the day you have to know what questions to ask of the recordings. We have to ensure that we are not replacing one kind of formulaic science with another. ■
This interview has been edited for clarity and length.
This post originally appeared on Medium
None of us would get on a plane that had its parts changed in mid-air, says Eve Marder, who has spent her career probing a very specific cluster of crustacean nerve cells. Yet we are all walking around undergoing a constant turnover of cellular parts, and so are the lobsters and crabs Marder studies. Within every nervous system, crucial components that mediate neural communication — receptors that detect chemical signals or pores that let ions in and out of neurons — are replaced hourly or daily, and the communication itself can be routed through various neurons. Marder studies how this complexity is maintained by artificially ‘crashing’ the nervous system.

The pyloric circuit, part of the group of nerve cells in the lobster that control digestion. The communication between these neurons is controlled by two neurotransmitters, acetylcholine (ACh in pink) and glutamate (Glu in blue). Image from Figure 4a of Marder & Bucher, 2007.
The clutch of lobster stomatogastric, or digestive, neurons is an ideal testbed for nervous system hacking: the small number of cells naturally maintain an exquisite temporal synchrony with each other but can be exposed to environmental extremes or modulated to produce configurations that drive the system to collapse. The orderly pattern of neural discharges then gives way to freakish rhythms or utter silence; these crashes reveal not just the variability that exists across cells and experimental animals but highlight the incredible robustness of the nervous system to external and internal shocks. Like an overheating laptop, the small network of cells can also shut down in high temperatures, though unlike a fried computer the cells can recover when the environment returns to normal.
How do nervous systems bounce back and maintain normal outputs in the face of constant internal change? This is the broader, career-spanning question for Marder, but her enthusiasm for hacking systems also extends from circuits to scientific publishing and beyond. During a recent visit to the RIKEN Brain Science Institute in Japan, the Brandeis professor and Kavli Prize winner talked to us about redundancy in the nervous system, her passion for open-access publishing, and why individual investigator grants are the way of the future.
“Connectomes are absolutely necessary but completely insufficient for understanding nervous system function”
How do people, or lobsters, maintain robust and functional behaviors while their nervous systems are constantly renewing and reconfiguring themselves?
Anything important the brain does, there will be more than one way of doing it. Circuit diagrams or connectomes are absolutely necessary but completely insufficient for understanding nervous system function. By studying degenerate mechanisms that produce the same behavior change we can probe the underlying parameter space, and I think this is a paradigm shift. We are not looking for one model that represents a mean of parameters. Rather, we try to build many models that cover the range of normal behavior parameters. The networks that we build may produce the same output with different parameter sets, but respond differently to perturbations. This kind of variability in animals is often hidden by compensating cellular mechanisms. How you can maintain a nervous system that is constantly breaking down and changing its parts, that for me is the deeper question.
“Knowledge should not be for sale”
As a Senior Editor at eLife, what does open access mean to you and are you seeing a shift, in both academics’ publishing choices and how peer review is conducted?
eLife is a rigorous journal that publishes roughly 15% of submitted papers, but we also have a philosophical stance on open access. For me personally, this came from my mother, who was interested in human brain development and in her later years was very deaf and consequently spent a lot of time online. She told me that academics forget that they have access, through their libraries, to all the world’s published research. But my mother, at home, didn’t have a university library. She said it’s criminal that she had to pay to access papers, because the abstract alone can’t tell you if you really want to read the paper or not. Fundamentally, knowledge should not be for sale. Most research, wherever it’s done, is funded through taxes, either directly by a government or indirectly through tax-privileged foundations and institutions. Working people, tax-paying people, should have rights to knowledge, and I feel strongly about that. Personally I will only publish in journals that have open-access options. Even commercial journals have options to make papers free six or twelve months after publication. Knowledge should not be privileged.
Researchers should also not be assessed based on impact factor — I know recent recruits who have gotten faculty jobs on the basis of papers in eLife or Journal of Neuroscience. Randy Schekman, eLife editor-in-chief, refuses to publicize eLife’s impact factor, though it is calculated by Web of Science, and it is quite respectable. eLife is a generalist journal and it has become a home for extraordinary papers that will become longstanding classics in their fields, such as the Drosophila lines or Homo naledi, because there is no limit on the number of figures or pages. These are papers with scope that some high-profile journals might be unwilling to publish. Some authors are sending their papers to eLife for other reasons, because they prefer the faster editorial process and know that we don’t ask for months of additional work to get a paper accepted.
High-profile journals have taught authors to oversell. This is the most common complaint I hear from reviewers: authors over-claiming novelty or generality, trying to make their result more important than it is.
“Brain tech development will make possible things that we could never have dreamed of”
How is the funding landscape changing for neuroscience?
I’ve been fortunate to have been funded by the NIH (US National Institutes of Health) throughout my career. The National Institute of Neurological Disorders and Stroke (NINDS) recently created a new grant mechanism,R35, which allows an investigator to consolidate several grants into a single multi-year project. Previously, in the US and working under NIH funding, you were tied to a specific project, and grants were awarded to projects not people. You had to keep strict track of which experiments belonged to which grant. This new award gives grants to people and is less restrictive. The model is more akin to that of the Howard Hughes Medical Institute or MacArthur Fellowships. When this change comes to pass, it will be a great relief for investigators to go in the direction the science takes us.
You were a member of the original BRAIN Initiative working group. What do you think the outlook is for neuroscience as the field becomes more global and collaborative?
The outlook is really positive. I’m not involved in the BRAIN Initiative anymore, but it was exciting to have been a part of its beginning, with 14 other very smart people who were all thinking unselfishly out of the box about what is best for the field. Everyone agreed that at that moment, new technologies that could be used by any investigator would be most useful, and that the best strategy was individual investigator awards and a bottom-up rather than top-down approach.
Last year at SfN, there were a number of talks from the first round of awards. Some really beautiful science was presented with interesting directions. This heyday of technology development is going to make possible things we could never have dreamed of. But at some point you have to transition from technology development to implementation. You may be recording from many neurons, but at the end of the day you have to know what questions to ask of the recordings. We have to ensure that we are not replacing one kind of formulaic science with another. ■
This interview has been edited for clarity and length.
neuroamanda
Science writer at the RIKEN Brain Science Institute in Japan
Blogging about brains on Medium